

Metronidazole : Side effects and Safety
Chaochen Wang$^1$, Yingsong Lin$^1$, Masumi Okuda$^2$, Shogo Kikuchi$^1$
1.Aichi Medical University School of Medicine 2.Hyogo College of Medicine
Background
Metronidazole (MNZ) has been broadly prescribed as therapy for \(H. pylori\) eradication worldwide.
Second line regimen using MNZ is covered under national health insurance in Japan.
Eradication rate :1
60.5% for PPI + AMPC + CLA (PAC) | 98.3% for PPI + AMPC + MNZ (PAM)Resistance rate of CLA in Japanese children is reported to be more than 40%.2
MNZ \(\Longrightarrow\) Possibly carcinogenic to human (2B)
ヒトに対する発がん性が疑われる
by International Agency for Research on Cancer, IARC in 1987
1: Unpublished data, Mabe et al.; 2: Kato and Fujimura 2010
Pubmed searching
Related literature (published until April. 2016) were reviewed.
Search term:
(("Drug-Related Side Effects and Adverse Reactions"[Mesh]) AND "Metronidazole"[Mesh]) OR ("Metronidazole/adverse effects"[Mesh] OR "Metronidazole/toxicity"[Mesh]) OR (("Metronidazole"[Mesh]) AND "Carcinogenicity Tests"[Mesh])
Evidence found | 動物実験の結果
Oral exposure of MNZ has shown carcinogenic activity in mice and rats.
| Results | Year | Authors | |
|---|---|---|---|
| Carcinogenic cites: | |||
| Pulmonary | \(\uparrow\) | 1972 | Rustia and Shubik |
| \(\uparrow\) | 1977 | IARC | |
| \(\uparrow\) | 1983 | Cavaliere et al. | |
| Liver | \(\uparrow\) | 1979 | Rustia and Shubik |
| Lymphomas | \(\uparrow\) | 1972 | Rustia and Shubik |
| Mammary-gland | \(\uparrow\) | 1979 | Rustia and Shubik |
| \(\uparrow\) | 1984 | Cavaliere et al. | |
| Pituitary-gland | \(\uparrow\) | 1979 | Rustia and Shubik |
| Genetic damage | Negative | 2000 | Touati et al |
| Reproductive Organs & Fertility |
\(\downarrow\) | 2013 | Kumari and Singh |
However,
not in humans
Lack of evidence for cancer due to use of metronidazole. N Engl J Med. 1979;301:519–522. ヒトでの追跡研究

Other cancer studies in Humans
Data on MNZ carcinogenecity for humans is still not sufficient.
No increased cancer risk in 12,000 users of MNZ.1 \(\Longrightarrow\) Only followed for 2.5 years. (A letter to JAMA)
No association between short-term exposure to MNZ and cancer in human were found in 5,222 MNZ user/nonuser pairs (RR 0.98; 95% CI, 0.80$-$1.20)2
Another retrospective study3 of children (\(<\) 5 \(y.\), n \(=\) 328,846) who had been exposed to MNZ in utero also reported negative results (RR 0.81; 95% CI, 0.41$-$1.59) on cancer incidence of all sites.
ヒトでの研究から,発がんリスクの上昇は認められていない.
Other potential side effects (まれな副作用)
- Brain/nerve related: seizures1 , aseptic meningitis2 , encephalopathy3 .
\(\Longrightarrow\) Prolonged use of MNZ. (脳神経系) - Gastrointestinal tract related: nausea, occasionally vomiting, diarrhea.4
\(\Longrightarrow\) Daily dose \(\geq\) 2250 mg. (消化器系) - Skin eruption5 , pancreatitis6 . (皮疹,膵炎)
- Drug-drug interaction:
- \(\uparrow\) the effect of warfarin.
- \(\downarrow\) the function of ALDH2.
1. Hari et al. 2013; 2. Farmakiotis et al. 2016; 3. Khan et al., 2007;4. Ohnishi et al. 2014;5. Kumar et al. 2013;6. O’Halloran et al. 2010.
Recommended regimens in UK (イギリスの小児除菌推奨レジメ)
| Age Range (\(y.\)) | Oral dose (mg per day) with omeprazole (PPI) |
combined with | ||
|---|---|---|---|---|
| AMPC | \(1\sim 6\) | 250, twice | CLA | |
| 125, 3 times | MNZ | |||
| \(6\sim 12\) | 500, twice | CLA | ||
| 250, 3 times | MNZ | |||
| \(12\sim18\) | 1000, twice | CLA | ||
| 500, 3 times | MNZ | |||
| CLA | \(1\sim12\) | 7.5 mg/kg (max. 500), twice | MNZ/AMPC | |
| \(12\sim18\) | 500, twice | MNZ/AMPC | ||
| MNZ | \(1\sim 6\) | 100, twice | CLA | |
| 100, 3 times | AMPC | |||
| \(6\sim 12\) | 200, twice | CLA | ||
| 200, 3 times | AMPC | |||
| \(12\sim18\) | 400, twice | CLA | ||
| 400, 3 times | AMPC |
Abbreviations: AMPC, amoxicillin; CLA, clarithromycin; MNZ, metronidazole.
Evidence-based guidelines from ESPGHAN and NASPGHAN for Helicobacter pylori infection in children. J Pediatr Gastroenterol Nutr. 2011;53:230–243.
北米・ユーロッパ小児除菌アルゴリズム:"
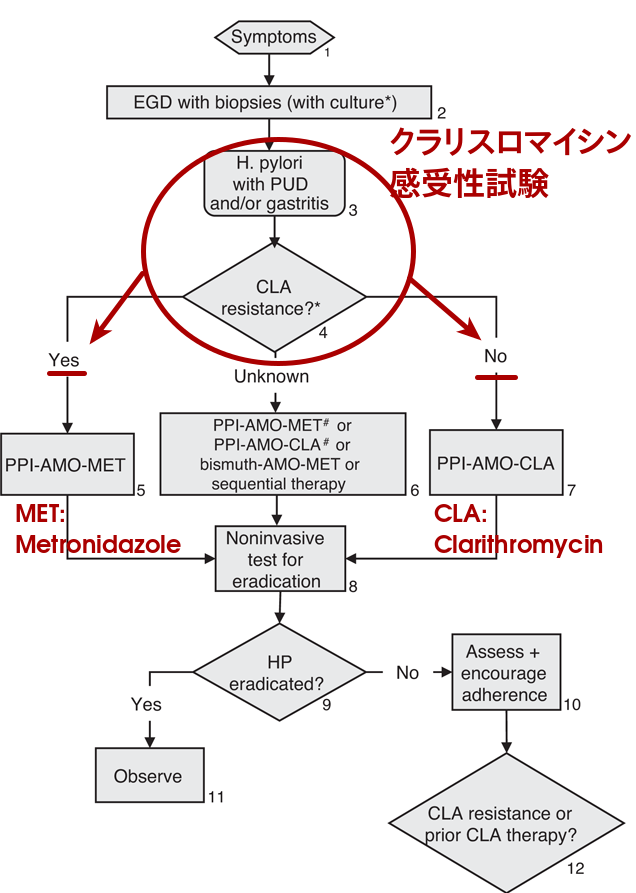
Conclusion
So far, there is no convincing evidence that short-term exposure to metronidazole would increase the risk of any cancer in human.
Considering the high resistance rate of clarithromycin in Japanese children, first-line therapy of PPI + amoxicillin + metronidazole would be an alternative option.
これまでの研究報告からみると,短期間の利用は,ヒトでのがん発生リスクの上昇は認められない.
日本の中高生においてクラリスロマイシンの感受性試験を行わない場合は,メトロニダゾールを含む併用療法は一つの選択肢となりうる.